Now, a team of researchers led by Salk Professor Susan Kaech has discovered that the cells responsible for long-term immunity in the lungs can be activated more easily than previously thought. The insight, published in the Journal of Experimental Medicine on June 11, 2020, could aid in the development of universal vaccines for influenza and the novel coronavirus.
“Inside our lungs exist long-lived killer T cells that recognize specific viruses and protect us against re-infection, should we encounter the virus again. Our results have elucidated the manner by which these cells ‘see’ the virus upon re-infection and provide rapid immunity,” says Kaech, director of Salk’s NOMIS Center for Immunobiology and Microbial Pathogenesis. “It also may help us understand long-term immunity as it relates to coronavirus.”
When we are first exposed to bacteria or viruses, such as influenza, one type of our immune cells, known as killer T cells, destroy infected cells to prevent the spread of the disease. Once the pathogen is cleared, these experienced killer T cells (also called killer “memory” T cells) remain in our body long-term, and “remember” previous invaders. These killer memory T cells enable our immune systems to more rapidly respond to a second attack and effectively provide long-term protective immunity against the invader, a fundamental concept behind vaccination.
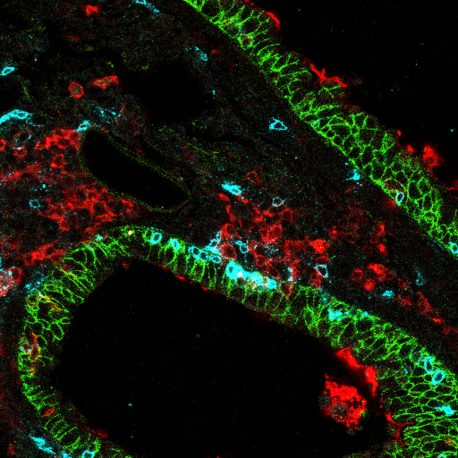
Lung-specific CD8 killer T cells (blue) in lung tissue (green) surrounded by dendritic lung cells (red).
Scientists know a lot about how killer memory T cells get activated in lymphoid organs (such as lymph nodes). Immune messenger cells called dendritic cells present fragments of the virus to the killer memory T cell, similar to a handler presenting a scent to a hound, to license their killer function.
But prior studies had not examined this interaction in vital organs, such as the lung. The lung is a frequent entry site for pathogens such as influenza and coronavirus, so the team set out to confirm whether this long-held dogma applied to killer memory T cells that reside in the lungs.
Kaech and then-graduate student Jun Siong Low, first author of the paper, assumed that dendritic cells would be required to reactivate killer memory T cells to fight a second viral attack. So, they deleted various types of messenger cells one at a time in mice to see if the killer memory T cells would still recognize a second influenza infection. The researchers used a green florescent reporter protein to make the killer memory T cells glow if they recognized the virus. However, each time the researchers deleted a specific cell type, the killer memory T cells in the lungs continued to glow.
“At first, our results were disappointing because it didn’t seem like our experiments were working; the killer memory T cells in the lungs continued to recognize the virus after the deletion of many different messenger cell types,” says Low, now a postdoctoral fellow at the Institute for Research in Biomedicine (IRB) at the Università della Svizzera Italiana, in Switzerland. “Soon, we realized that these lung-resident killer memory T cells were special because they were not reliant on any single type of messenger cell. Instead, they could ‘see’ the second influenza infection through a variety of different messenger cells, including non-immune cells like lung epithelial cells, which was a remarkably exciting finding.”
In contrast, when the researchers examined the killer memory T cells in the lymph nodes—glands that swell during infections—they found that the killer memory T cells needed dendritic cells to recognize the second viral attack. This suggests that the anatomical location of the killer memory T cells dictates how they get reactivated, challenging the long-held dogma that killer memory T cells require dendritic cells for reactivation. The results help to reshape the paradigm of killer memory T cell activation.
Because lung-resident killer memory T cells can be quickly reactivated by nearly any cell type at the site of pathogen entry, identifying vaccines that can create these lung-resident killer memory T cells will likely be critical for superior immunity to viral infections of the lungs.
“We will take this knowledge into our next study, where we will examine whether lung-resident killer memory T cells form after a coronavirus infection,” says Kaech, holder of the NOMIS Chair. “Since not all infections induce killer memory T cells, we will determine if these cells form after a coronavirus infection and whether they can be protective against future coronavirus infections.”
Other authors included Yagmur Farsakoglu of Salk; Esen Sefik, Christian C.D. Harman, Ruaidhri Jackson, Justin Shyer, Xiaodong Jiang, and Richard A. Flavell of the Yale University School of Medicine; Maria Carolina Amezcua Vesely of the Universidad Nacional de Córdoba, in Argentina; Joseph B. Kelly of Stony Brook University and Linda S. Cauley of the University of Connecticut Health Center.
The work was supported by the NOMIS Foundation; the National Institutes of Health (R01 AI123864, R37 AI066232, S10 OD020142, P30 CA106359-39); A*STAR National Science Scholarship PhD; a Swiss National Science Foundation Early Postdoc Mobility Fellowship (P2BEP3_178444); a George E. Hewitt Foundation fellowship; the Howard Hughes Medical Institute; the Yale Center for Research Computing; the Yale Center for Genome Analysis; and the Waitt Advanced Biophotonics Core at Salk Institute for Biological Studies.
salk.edu


