In order to better understand how the blood-brain barrier protects our central nervous system from harmful substances in the bloodstream, ETH researchers have developed a new in-vitro model. (Image: Gunilla Elam / Science Photo Library
Mario Modena is a postdoc working in the Bio Engineering Laboratory at ETH Zurich. If he were to explain his research on the blood-brain barrier – the wall that protects our central nervous system from harmful substances in the blood stream – to an 11-year-old, he would say: “This wall is important, because it stops the bad guys from getting into the brain.” If the brain is damaged or sick, he says, holes can appear in the wall. Sometimes, such holes can actually be useful, for example, for supplying the brain with urgently needed medicine. “So what we are trying to understand is how to maintain this wall, break through it and repair it again.”
This wall is also important from a medical perspective, because many diseases of the central nervous system are linked to an injury to the blood-brain barrier. To discover how this barrier works, scientists often conduct experiments on live animals. In addition to such experiments being relatively expensive, animal cells may provide only part of the picture of what is going on in a human body. Moreover, there are some critics, who question the basic validity of animal testing. An alternative is to base experiments on human cells that have been cultivated in the laboratory.
Cell-cell communication largely overlooked
The problem with many in-vitro models is that they recreate the blood-brain barrier in a relatively simplified way using blood-vessel-wall cells (endothelial cells). This approach fails to represent the complex structure of the human system and disregards, for instance, the communication between the various cell types. Furthermore, many of these models are static. In other words, the cells are floating in a suspension that is not moving, which implies that fluid flow or the shear stress the cells are exposed to in the body are not considered.
There are also dynamic in-vitro models that simulate flow conditions in the body, but the catch here is that the pumps they require make the experimental setup rather complicated. Alongside all these challenges, there is the problem of measurement: it is all but impossible to take high-resolution images of structural changes to the blood-brain barrier in real time while also measuring the barrier’s electrical resistance, both of which reflect barrier compactness and tightness.
Killing several birds with one stone
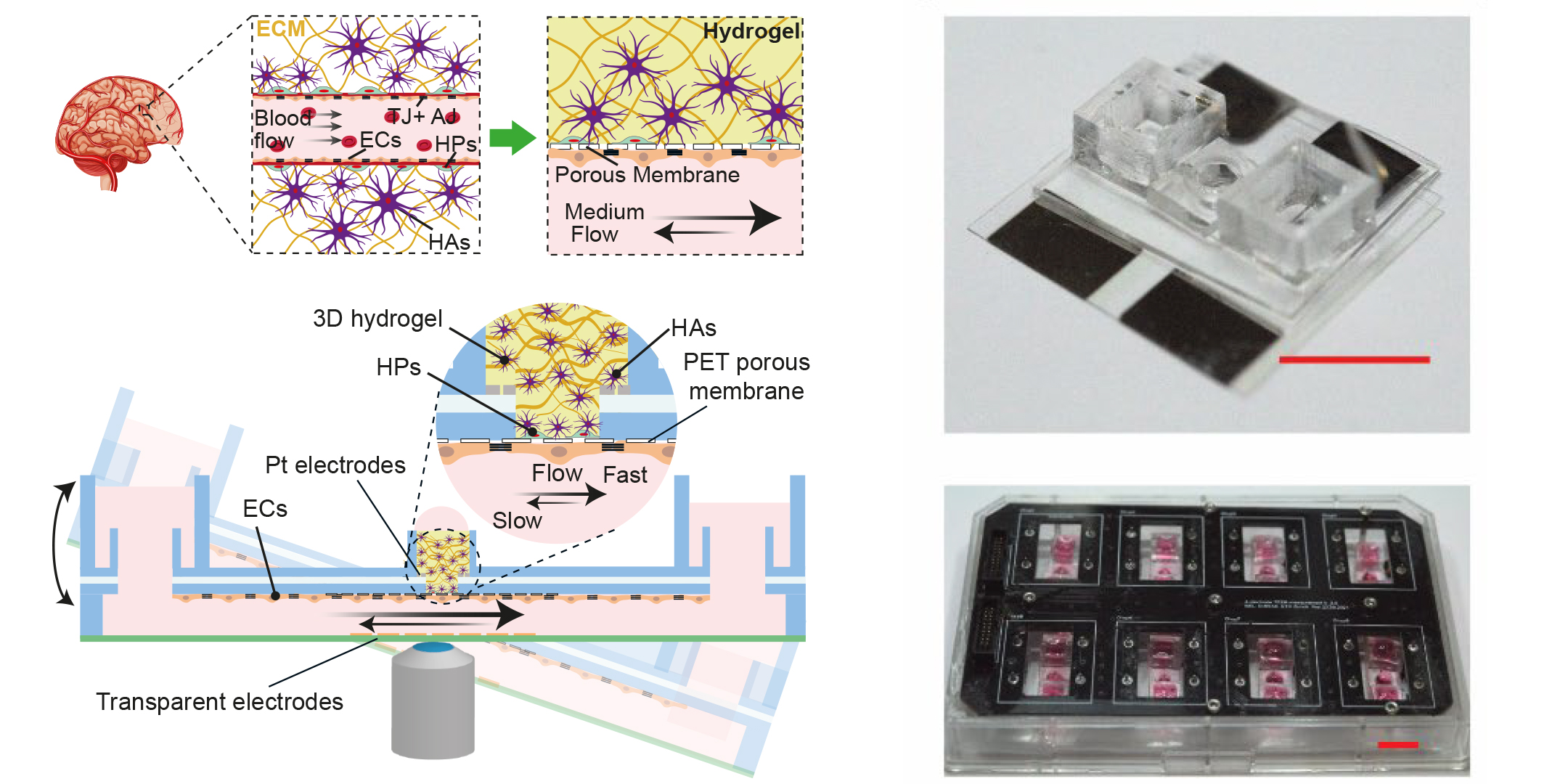
If each of these challenges were a bird, Modena’s platform would be the proverbial stone that kills them all. Working under Andreas Hierlemann, Modena and his colleagues spent three and a half years developing the open-microfluidic 3D blood-brain barrier model.
To recreate the barrier, the research team took those cell types that naturally make up the blood-brain barrier – microvascular endothelial cells, human astrocytes and human pericytes – and combined them within a single platform. “This strategy allowed us to almost fully replicate the 3D cell structure found in the human body,” Modena says. “But what’s really exceptional is that we can measure the barrier’s permeability while simultaneously mapping morphological changes to the barrier by means of high-resolution time-lapse microscopy.” To facilitate this double act, the researchers deposited entirely transparent electrodes on glass coverslips on both sides of the barrier to measure its permeability, which is reflected in the electrical resistance across the cell barrier. Transparent electrodes offer a decisive advantage over other types of electrodes, which include metal films or wire structures that may interfere with optical detection and high-resolution microscopy.
“Without increasing the complexity”
To mimic the way fluid flows in the body, the researchers realized the microfluidic platform with fluid reservoirs at both ends on a kind of seesaw. Gravity then triggered the flow, which - in turn - generated shear force on the cells. Hierlemann explains the benefit of this setup: “Since we are not using any pumps, we can experiment with multiple model systems simultaneously, for instance in an incubator, without increasing the setup complexity.”
In a study, published recently in the journal Advanced Science, the researchers presented and tested their new in vitro blood-brain barrier model. They subjected the barrier to oxygen-glucose deprivation, as happens when someone is having a stroke. “These experiments allowed us to trigger rapid changes in the barrier and demonstrate the platform’s potential,” Modena says.
Pharma company already showing an interest
Through this study, Modena and his colleagues were able to do more than showing that their new platform is suitable for taking measurements. They also discovered that the barrier’s electrical resistance decreases even before it undergoes morphological changes that make it more permeable. “This finding could prove relevant for future research,” Modena says. The team also observed that in control experiments using a static in-vitro model, the barrier was more permeable than in the new dynamic setup. “It is clear that the shear force, generated by the gravity-driven flow, promotes the formation of a denser barrier layer, which confirms how important flow is for representative in-vitro models” Modena says.
Modena and Hierlemann believe that their model will make it easier to detect which molecules stabilise the barrier, as well as to discover compounds and methods suitable for crossing it, which would be useful in the treatment of brain tumours. But Hierlemann notes that the model could also change the course of future in-vitro research: “The advantage of our platform is that it is very easy to adapt to other endothelial cell models, where a combination of barrier-tightness measurements and high-resolution microscopy could pave the way to new research.” Industry has manifested interest in the new the model. A pharmaceutical company is already in contact with the researchers.
Vinzenz Greiner
ethz.ch


